 So, this makes the ion have a negative charge. Predicting charges on monatomic cations and anions Did you know that you can use the periodic table to predict the charges certain elements will have when they. When we gain more electrons, we can know that the amount of electrons is higher than the proton. 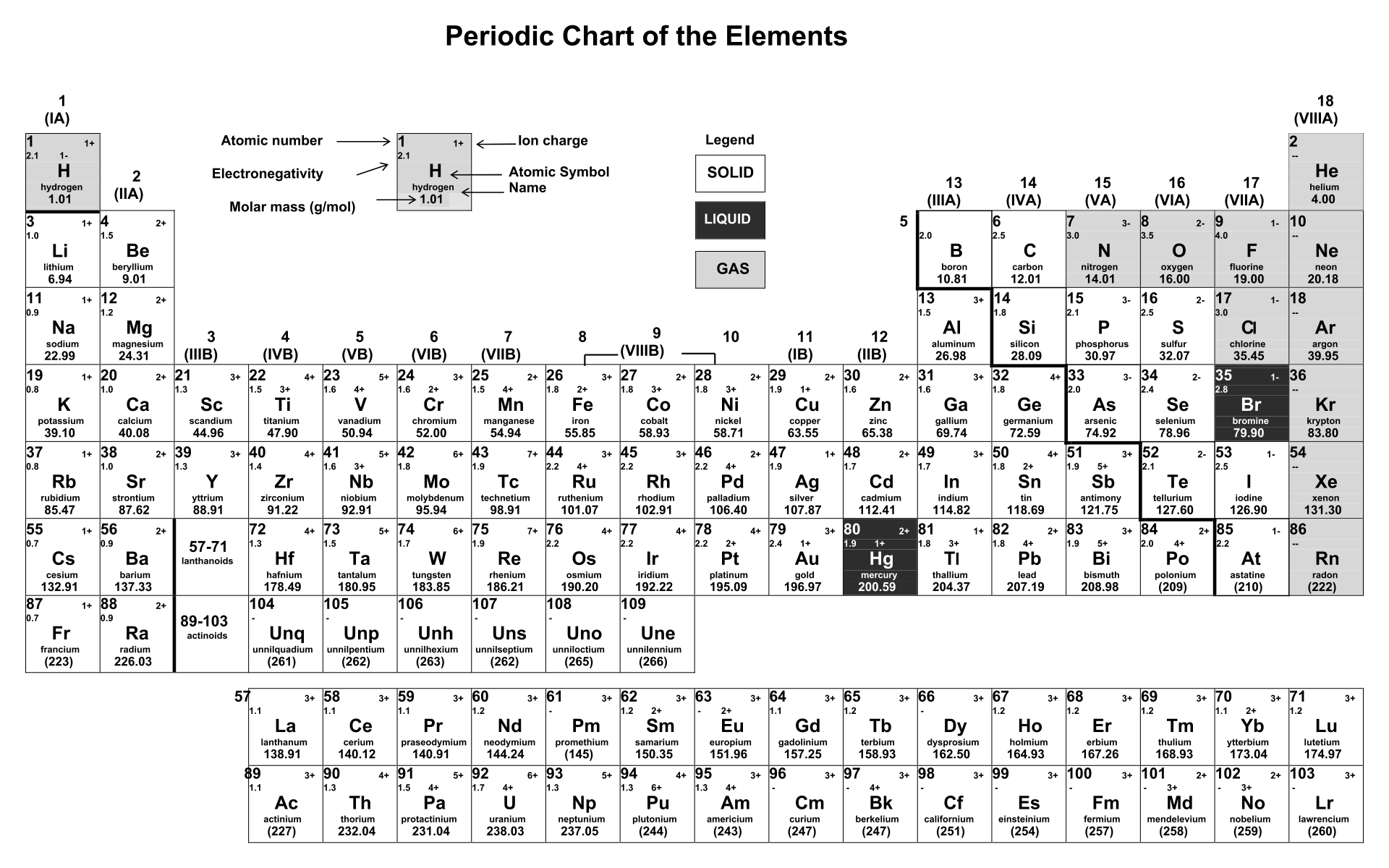
So, we can conclude that this ion has a positive charge and vice versa. So, the dominant charge of the certain ion is the proton. If the ion loses the electron, this means that the negative charge is lesser than the positive charge (proton). Elements on the left side of the periodic table, metals, lose the electrons necessary to reach the electron configuration of the nearest noble gas. It is a depiction of the periodic law, which says that when the elements are arranged in order of their atomic numbers an approximate recurrence of their. Actually, to understand the ion, we can see how the atoms lose their negative charge (electron). The elements on the right side of the periodic table, nonmetals, gain the electrons necessary to reach the stable electron configuration of the nearest noble gas. Actually, we have discussed this before above. When we download a periodic table of ions, we also need to know about how to determine an ion. Then, if it is reversed, which means the amount of electrons is higher than the proton, we can call it anion. The cation is where the positive charge (proton) has more amount than the electron. There are two kinds of ions in the periodic table of ions. Ions are single charged atoms (simple ions) or small charged molecules (polyatomic ions). However, it is pretty normal to be found that there is an imbalance of the charge, whether the number of protons is higher or the number of electrons is higher. Well, there are two kinds of ions on the periodic table.Īctually, if an atom or atoms that have equal numbers of negative charge (electrons) and positive charge (proton), then they can be defined as a neutral atom. Yes, one of the things that is required to be understood in the beginning is the ions themselves. 
Yes, we can call this a periodic table of ions.īefore we download the periodic table of irons, we actually need to understand the basic things inside the periodic table of ions. When it comes to learning Chemistry lessons, we may have seen our teacher introduce us with a sheet of the grid that consists of many abbreviations as a symbol of the elements. In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
